*Corresponding Author:
Ali N,
Department of Cardiology, Leeds General Infirmary, Great George Street, Leeds, LS 3EX, UK
Tel: 0113 2432799
E-mail: nomanali456@doctors.org.uk
Abstract
Aims: Refractory Angina (RA) is characterised by persistent anginal symptoms despite optimal medical therapy and revascularization. The aim of this study is to assess the effects of Coronary Sinus ReducerTM (CSR) implantation on symptom burden and quality of life in patients with RA.
Methods: This single-centre retrospective study assessed effectiveness of CSR implantation at improving anginal symptoms using preand post-implantation Canadian Cardiovascular Society (CCS) scores, as well as quality of life using Short Form-36 (SF-36) and Hospital Anxiety and Depression Scale (HADS) scores.
Results: 18 patients with a mean age of 64 years (SD 9.0) underwent CSR implantation. The majority had undergone prior revascularisation, either via Percutaneous Coronary Intervention (PCI), Coronary Artery Bypass Grafting (CABG) or both (83%; 15/18). The patients had preserved left ventricular systolic function, with a mean ejection fraction of 56% (SD 9.2). The median number of anti-anginal medications prescribed per-patient was 4 (IQR 3-5). Significant improvements were noted in CCS (2.8 vs. 2.0; p<0.01) and SF-36 scores (40.4 vs. 54.1; p<0.01) after 21 months median follow-up. All procedures were successfully performed without major complications.
Conclusion: Our study demonstrates CSR implantation to be safe and associated with significant improvements in anginal symptoms and quality of life.
Keywords
Anginal symptoms; Coronary sinus; Quality of life; Re- fractory angina
Introduction
Refractory angina (RA) is a chronic condition characterized by persistent anginal symptoms despite Optimal Medical Therapy (OMT) and revascularization. In order to fulfill the conventional definition of the disease, coronary insufficiency in the setting of Coronary Artery Disease (CAD) needs to be demonstrated, and reversible myocardial is chaemia should be clinically established as the cause of the symptoms [1].
Whilst definitive epidemiological data pertaining to the prevalence of RA is scarce, there is a general recognition that it is a growing problem and one that results in a significant public health burden [2]. The refractory and debilitating nature of the symptoms associated with the condition can result in a marked detriment to quality of life, with sufferers often requiring recurrent hospital admissions.
Moreover, patients may develop a maladaptive psychological response to their disorder, which has exacerbating effects on utilization of services in the secondary care setting [3]. Following the publication of data demonstrating long-term mortality of patients with RA to be lower than previously reported [4],as well as the fact that patients with RA preserve their left ventricular systolic function despite frequent is chaemic episodes [5], in recent years the focus of treatment has shifted towards alleviation of symptoms and improved quality of life. However, achieving these aims can be challenging in this cohort of patients due to the complex interplay between physical and psychological factors in RA. As such, patients often require treatment modalities beyond the traditional cornerstones of pharmacotherapy and coronary revascularisation. Increasingly, psychological interventions such as Cognitive-Behavioural Therapy (CBT) and education programmes are being used to good effect amongst patients with RA. Indeed our group has previously published data demonstrating that this intervention has a sustainable impact on improving quality of life for patients with RA [6].
Nevertheless, despite the above interventions, a subset of patients remains persistently symptomatic. In recent years, a novel therapy has become available for patients with RA who have no further revascularization options, and who demonstrate inducible ischaemia on functional testing: Coronary Sinus ReducerTM (CSR; Neovasc Inc) implantation. This hour-glass shaped, balloon-expandable, stainless-steel device is percutaneously implanted into the coronary sinus via right internal jugular venous access (Figure 1). It produces a local narrowing of venous drainage which results in increased upstream pressure within the sinus. This is postulated to redistribute collateral flow from non-ischaemic to underperfused, ischaemic regions of myocardium. COSIRA was a multi-centre, prospective, randomized, double-blinded trial to assess safety and efficacy of the CSR [7]. Patients were included if they had symptoms indicative of RA (Canadian Cardiovascular Class [CCS] class III or IV) despite optimal therapy and revascularization, with confirmed reversible ischaemia in anterior or lateral wall of the left ventricle. In comparison with a sham procedure, CSR implantation was associated with significant benefit to anginal symptom burden and quality of life at 6-month follow-up [7].
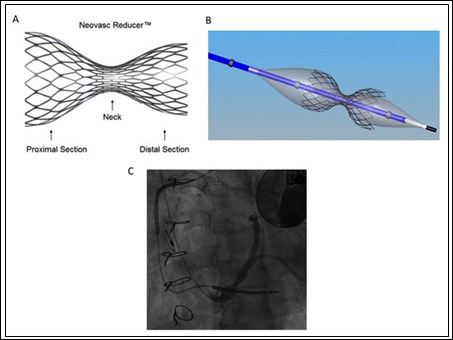
Figure 1: Coronary sinus reducer. A: An illustration demonstrating the ‘hour-glass’ shape of the stent; B: An illustration of the stent crimped onto a balloon, prior to deployment; C: An angiographic image of the stent deployed within the coronary sinus. Images taken with permission from Professor Shmuel Banai.
Our centre is one of only [5] in the UK that performs CSR implantation for RA. The aim of the present study is to provide the first real-world dataset from the UK on the safety and efficacy of CSR implantation, focusing on the effect on anginal symptom burden and quality of life.
Methods
This study was carried out as a single-centre, retrospective analysis of patients who underwent CSR implantation. The decision to implant a CSR was made by the cardiology team following a multi-disciplinary review of patients’ symptoms, medication regimens, results from functional imaging, and revascularisation options. The eligibility criteria for CSR implantation reflect that from the COSIRA trial, and were as follows:
- Persistent anginal symptoms despite maximally-titrated medical
- Reversible ischaemia within the left coronary territory, demonstrated by myocardial perfusion
- Proven CAD on angiography not amenable to percutaneous coronary intervention (PCI) or coronary artery bypass grafting (CABG).
Exclusion criteria included recent PCI (≤ 6 months), acute coronary syndrome (≤ 3 months) or presence of pacemaker leads within the right heart. Following implantation, patients were maintained on dual antiplatelet therapy with aspirin and clopidogrel for one month.
Data on the characteristics of patients who underwent CSR implantation was collected retrospectively using electronic records and information on patient demographics, coronary anatomy, medication regime, history of revascularisation and functional imaging results were collated.
Prior to device implantation, patients were graded on the CCS scoring system to quantify level of anginal symptoms. They then went on to complete well-established and validated questionnaires, providing a quantitative measure of quality of life as well as levels of anxiety and depression. The Short-Form Health Survey (SF-36) was used to assess health status [8]. It consists of eight scale scores, each carrying equal weight. Areas covered include vitality, physical functioning, bodily pain, general health perceptions, physical, emotional and social role functioning, and mental health. A score of 0 constitutes maximum disability, with 100 equivalents to no disability. The Hospital Anxiety and Depression Scale (HADS) were used to as a measure of anxiety and depression [9]. It incorporates a seven-item scale for each, with a score ranging from 0-3. Therefore, a person can score between 0 and 21 for both, giving a combined total maximum score of 42. CCS, SF-36 and HADS scoring was repeated at least 6 months post-procedure via telephone consultation with one of three co-authors, none of whom were involved in the process of device implantation, and all of whom were blinded to the pre-implantation scores. The efficacy of CSR implantation was assessed via comparison of pre-and post-implantation scores, and analysis of peri and post-procedural complications was undertaken to ascertain the safety of the intervention.
Data collation and statistical analyses were performed using Microsoft Excel for Mac 2011. Continuous variables are presented as means with Standard Deviations (SD). Parametric data were compared using 2-tailed, paired student’s t-test, whilst the Wilcoxon Signed-Rank test was used for non-parametric data. P values of less than 0.05 were deemed to be of statistical significance.
Results
The study included 18 patients (Table 1) who underwent CSR implantation between January 2013 and November 2016. Of these, 2 were included in the COSIRA trial. The majority was male (78%; 14/18) and had undergone prior revascularisation, either via PCI, CABG or both (83%; 15/18). At the time of CSR implantation the mean age of this cohort was 64 years (SD 9.0), whilst the median number of anti-anginal medications prescribed per-patient was 4 (IQR 3-5). In addition to traditional anti-anginal agents such as beta blockers, calcium channel blockers and nitrates, the majority of these patients were also taking adjunctive pharmacotherapy prior to CSR implantation. All patients were trialled on ranolazine, and 83% (15/18) tolerated the drug long-term. The remaining 3 patients discontinued the drug due to side-effects. Half of the patients (50%; 9/18) were prescribed ivabradine, and all tolerated it. Only 2 patients were on neither ranolazine nor ivabradine at the time of CSR implantation. Results of myocardial perfusion testing pre-implantation confirmed that this cohort had generally preserved Left Ventricular Systolic Function (LVSF), with a mean ejection fraction of 56% (SD 9.2).
The median time between device implantation and follow-up questionnaire was 21 months (IQR12-32). A significant improvement was noted in mean CCS score (2.8[0.6] vs. 2.0[0.8]; P=0.002) (Figure 2). The majority of patients reported an improvement of at least 1 CCS category (11/18; 61%) and none were found to have a worsened CCS score. A significant improvement was also noted in SF-36 scores (40.4[18.9] vs. 54.1[18.7]; P=0.0005) (Figure 3). No significant difference was noted in HAD scores (17.3[7.6] vs. 14.1[9.8]; P=0.14) ( Figure 4)
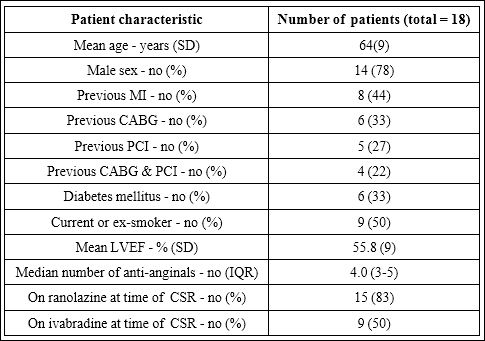
Table 1: The characteristics of patients receiving a coronary sinus reducer.
CABG: Coronary Artery Bypass Grafting; IQR: Interquartile Range; LVEF: Left Ventricular Ejection Fraction; MI: Myocardial Infarction; PCI: Percutaneous Coronary Intervention.
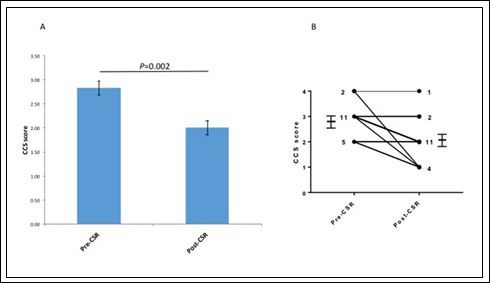
Figure 2: Comparison of Anginal Symptom burden (CCS score) preand post-implantation of Coronary Sinus Reducer (CSR). A: Comparison of mean CCS scores; error bars represent SEM. B: Comparison of individual CCS scores; numbers denote patients in each scoring category (n=18).
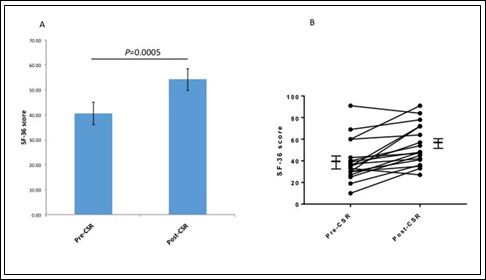
Figure 3: Comparison of quality of life score (SF-36) preand post-implantation of Coronary Sinus Reducer (CSR). A: Comparison of mean SF-36 scores; error bars represent SEM. B: Comparison of individual SF-36 scores (n=18).
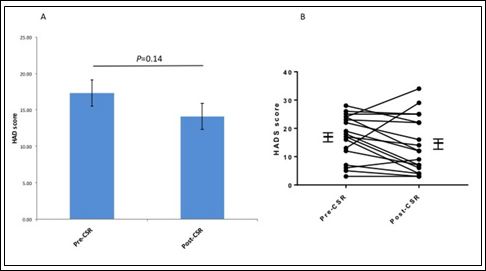
Figure 4: Comparison of anxiety and depression (HADS) preand post-implantation of coronary sinus reducer (CSR). A: Comparison of mean HAD scores; error bars represent SEM. B: Comparison of individual HAD scores (n=18).
The 18 patients included in this study encompassed the operator’s learning curve for CSR implantation. Peri-procedural complications were noted in 4 cases (22%), with small coronary sinus dissections in 2 patients and 2 minor stent displacements at the time of deployment.
The coronary sinus dissections caused no haemodynamic effects, and the patients were managed as normal following implantation. The stent displacements occurred following balloon inflation due to irregularity within the sinus. In spite of the minor displacements from the intended location, the final stent positions were acceptable in both cases and so no further intervention was required. All 18 patients included in this study have remained complication-free post-procedure.
Discussion
The present study evaluated the effect of CSR implantation on anginal symptom burden and quality of life in patients with RA, inducible ischaemia and no remaining coronary revascularisation options. It provides the first real-world outcome data from a UK centre on this subject.
Our study demonstrates CSR implantation to be associated with a significant improvement in anginal symptom burden after a median follow-up period of 21 months. This finding is consistent with those from previously published studies [7,10,11]. We also went on to demonstrate a significant improvement in quality of life, as measured by the SF-36 scoring system. Overall, our results align closely with those from a recently published retrospective analysis from an Italian centre [12]. Analysis of the characteristics of patients who underwent CSR implantation in the present study demonstrates that the majority is male, has generally preserved LVSF, is on multiple anti-anginal medications and has previously undergone coronary revascularisation. These observations are broadly consistent with demographic data derived from the patient cohort included in the pivotal COSIRA trial [7].
Whilst the use of a CSR as a treatment for RA is relatively novel, the underlying concept underpinning it is not; improvement in anginal symptoms associated with ligation of great cardiac veins was described as far back as 1941 by the Canadian surgeon Mercier Fauteux [13]. Further refinement of the concept was seen in 1954, when Beck and Leighninger reported improvements in anginal symptoms and functional status associated with partial occlusion of the coronary sinus following surgical ligation [14-18] (see ‘The history of myocardial revascularization before the advent of cardiopulmonary bypass’ for a detailed historical perspective [19]). The physiological basis for improvement in anginal symptoms via narrowing of the coronary sinus remains incompletely understood. It is thought that patients with CAD develop dysfunction of a sympathetically-mediated vasoconstrictive response in sub-epicardial vessels, which would otherwise serve to increase blood flow to sub-endocardial capillaries during periods of heightened myocardial oxygen demand [20]. It is postulated that the resulting sub-endocardial ischaemia is contributory to the pathogenesis of RA [21]. Canine studies have demonstrated that increasing coronary venous pressure by sinus occlusion assists in redistributing blood from the less ischaemic epicardium to endocardium, thus reducing myocardial ischaemia [21]. Interestingly, this process takes a number of months since it is contingent upon endothelialisation of the stent [20]. As such, we counsel patients that they are unlikely to feel any effects from the device for at least 3 months.
Another potential mechanism for the beneficial effects of CSR implantation pertains to increased venous pressure driving formation of collateral vessels within the ischaemic myocardium via intra-myocardial and epicardial angiogenesis [22]. This mechanism was postulated based upon histological analysis of myocardium following insertion of a small balloon pump into the coronary sinus of patients, post-infarct. However, this hypothesis remains contentious [23]. Whilst the exact mechanisms of action remain an area of uncertainty, taken together these hypotheses provide a putative explanation for the findings of subsequent clinical trials which have demonstrated a significant reduction in inducible ischaemia following CSR implantation [2,10].
The finding of improved anginal symptoms and quality of life amongst patients already taking a multitude of anti-anginal medications is encouraging, particularly given that CSR implantation appears to be a safe and well tolerated therapeutic intervention; despite this series of patients incorporating the operator’s learning curve for the procedure, no complications of haemodynamic or long-term consequence were noted.
It is important to note that a number of other treatment modalities exist for patients with RA, some of which have the benefit of being non-invasive and do not require exposure to ionizing radiation. Examples include Extracorporeal Shockwave Myocardial Revascularization therapy (ESMR) [24,25] and Enhanced External Counterpulsation (EECP) [26]. Nevertheless, CSR implantation appears to be a highly effective therapy for patients with RA, even when compared to other treatment modalities [27].
The present study has certain limitations which must be borne in mind when appraising the results. It is a single-centre study with small sample sizes, and data was predominantly acquired retrospectively. This imbues certain limitations which are inherent to this type of research, such as the inability to account for confounding factors and the dependence on previously acquired data which may be of variable quality and validity. Furthermore, no control group was available for comparison to the patients who underwent intervention, precluding the ability to distinguish therapeutic effect from placebo. Outcome data was predominantly derived from responses to surveys and questionnaires, which are inherently subjective. Moreover, data was accrued using telephone consultations, and this methodology is susceptible to the risk of indirect coercion. Finally, the fact that postCSR measures were collated at varying time-points following implantation introduces a level of heterogeneity into the results that may compromise external generalizability. Nevertheless, whilst keeping these caveats in mind, the fact that our findings convincingly corroborate those from COSIRA7, a blinded, randomized, placebo-controlled trial, provides validity to our observations.
Conclusion
RA is a chronic disease that is associated with debilitating morbidity and compromised quality of life. Whilst medical therapy with first line anti-anginal agents remains the cornerstone of treatment, it is often challenging to establish patients on OMT due to side effects and/ or haemodynamic compromise. For patients who remain persistently symptomatic, and have demonstrable inducible ischaemia, CSR implantation has recently become a viable option. Existing literature provides encouraging data to support its use in this cohort of patients. The present study provides the first UK-derived real-world outcome data for such patients, and demonstrates CSR implantation to be associated with significant improvements in anginal symptoms and quality of life. Based on these findings, further prospective trials with larger cohorts of patients are warranted, and we anticipate that the results of the ongoing REDUCER-I registry will provide further evidence with which to assess its efficacy in a real world population [28].
References
- Mannheimer C, Camici P, Chester MR, Collins A, DeJongste M, et al. (2002) The problem of chronic refractory angina; report from the ESC Joint Study Group on the Treatment of Refractory Eur Heart J 23: 355-370.
- Banai S, Ben Muvhar S, Parikh KH, Medina A, Sievert H, et al. (2007) Coronary sinus reducer stent for the treatment of chronic refractory angina pectoris: a prospective, open-label, multicenter, safety feasibility first-in-man J Am Coll Cardiol 49: 1783-1789.
- Moore RK, Groves DG, Bridson JD, Grayson AD, Wong H, et al. (2007) A brief cognitive-behavioural intervention reduces hospital admissions in refractory angina J Pain Symptom Manage 33: 310-316.
- Henry TD, Satran D, Hodges JS, Johnson RK, Poulose AK, et al. (2013) Long-term survival in patients with refractory angina. Eur Heart J 34: 2683-
- Slavich M, Maranta F, Fumero A, Godino C, Giannini F, et al. (2016) LongTerm Preservation of Left Ventricular Systolic Function in Patients With Refractory Angina Pectoris and Inducible Myocardial Ischemia on Optimal Medical Am J Cardiol 117: 1558-1561.
- Patel PA, Khan M, Yau C, Thapar S, Taylor S, et al. (2016) The shortand long-term impact of psychotherapy in patients with chronic, refractory angi Br J Cardiol 23: 57-60.
- Verheye S, Jolicoeur EM, Behan MW, Petterson T, Sainsbury PA, et al. (2015) Efficacy of a device to narrow the coronary sinus in refractory N Engl J Med 372: 519-27.
- RAND Health. 36-Item Short Form Survey from the RAND Medical Outcomes
- Zigmond AS, Snaith R (1983) The hospital anxiety and depression scale. Acta Psychiatr Scand 67: 361-370.
- Konigstein M, Meyten N, Verheye S, Schwartz M, Banai S (2014) Transcatheter treatment for refractory angina with the Coronary Sinus Reducer. EuroIntervention 9: 1158-1164.
- Abawi M, Nijhoff F, Stella PR, Voskuil M, Benedetto D, et al. (2016) Safety and efficacy of a device to narrow the coronary sinus for the treatment of refractory angina: A single-centre real-world experience. Neth Heart J 24: 544-551.
- Giannini F, Baldetti L, Ponticelli F, Ruparelia N, Mitomo S, et al. (2018) Coronary sinus reducer implantation for the treatment of chronic refractory angi- na: a single-center JACC Cardiovasc Interv 11: 784-792.
- Sánchez-Borges M, González-Aveledo LA (2011) Photoallergic reactions to angiotensin converting enzyme inhibitors. J Eur Acad Dermatol Venereol 25: 621-622.
- Fauteux M, Palmer JH (1941) Treatment of Angina Pectoris of Atheromatous Origin by Ligation of the Great Cardiac Can MedAssoc J 45: 295-298.
- Beck CS, Leighninger DS (1954) Operations for coronary artery disease. J Am Med Assoc 156: 1226-1233.
- Beck CS, Leighninger DS (1955) Scientific basis for the surgical treatment of coronary artery J Am Med Assoc 159: 1264-1271.
- Sandler G, Slesser BV, Lawson CW (1967) The Beck operation in the treatment of angina Thorax 22: 34-37.
- Wising PJ (1963) The Beck-1 operation for angina pectoris: medical Acta Med Scand 174: 93-98.
- Brofman BL (1956) Medical evaluation of the Beck operation for coronary artery J Am Med Assoc 162: 1603-1606.
- Crea F, Galassi AR, Kaski JC, Nijhoff F, Lakemeier MDM et al. (1989) Effect of theophylline on exercise-induced myocardial Lancet 1: 683-686.
- Picichè M (2012) The history of myocardial revascularization before the advent of cardiopulmonary bypass. In: Picichè M (ed.). Dawn and Evolution of Cardiac Procedures: Research Avenues in Cardiac Surgery and Interventional Springer-Verlag: Heidelberg 65-77.
- Benedetto D, Abawi M, Stella PR, Nijhoff F, Lakemeier MDM, et al. (2016) Percutaneous Device to Narrow the Coronary Sinus: Shifting Paradigm in the Treatment of Refractory Angina? A Review of the Literature. Front Cardiovasc Med 3:
- Ido A, Hasebe N, Matsuhashi H, Kikuchi K (2001) Coronary sinus occlusion enhances coronary collateral flow and reduces subendocardial Am J Physiol Heart Circ Physiol 280: 1361-1367.
- Mohl W, Glogar DH, Mayr H, Losert U, Sochor H, et al. (1984) Reduction of infarct size induced by pressure-controlled intermittent coronary sinus occlu Am J Cardiol 53: 923-928.
- Paz Y, Shinfeld A (2015) Re: “Transcatheter treatment for refractory angina with the coronary sinus reducer” by Maayan Konigstein et al. EuroIntervention 11: 727-728.
- Slavich M, Pizzetti G, Vella AM, Carlucci C, Margonato D, et al. (2018) Extracorporeal myocardial shockwave therapy; a precious blast for refractory angina Cardiovasc Revasc Med 19: 263-267.
- Slavich M, Ancona F, Margonato A (2015) Extracorporeal shockwave myocardial revascularization therapy in refractory angina patients. Int J Cardiol 194:
- Ali N, Jamil HA, Waleed M, Raheem O, Patel P, et (2018) The effect of Enhanced External Counterpulsation on functional exercise capacity and symptom burden in patients with refractory angina. Br J Cardiol 011.
- Slavich M, Giannini F, Godino C, Pizzetti G, Gramegna M, et al. (2018) Reducer, extracorporeal shockwave therapy or stem cells in refractory angina: a retrospective J Cardiovasc Med (Hagerstown) 19: 42-44.
- REDUCER-I: (2016) An Observational Study of the Neovasc Reducer™
Citation: Ali N, Patel PA, Jamil H, Waleed M, Thapar S, et al. (2018) The Coronary Sinus Reducer™ System for the Treatment of Patients with Refractory Angina: A Single Centre UK Experience. J Cardio Cardiovasu Med 3: 009.
Copyright: © 2018 Ali N, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.


