*Corresponding Author:
Dejenie Mengistie,
Addis Ababa University, Ethiopia
E-mail: dejeniebiot2006@gmail.com
Abstract
In vitro meat production is the production of meat outside the food animals by culturing the stem cells derived from farm animals using scientific innovations and technological advancements. The production involves two techniques, self-organize and scaffolding techniques. The aim of this review is to provide scientific information about lab-growing meat production from stem cells within the bioreactor, and to see their advantage, disadvantage, and future prospects. Many issues arise on the conventional meat production like (excessively brutal slaughter of food animals, nutrition-related diseases, meat quality, food borne illnesses, resource use, antibiotic-resistant pathogen strains, and massive emissions of methane that contribute to global warming), from animal rights activists. A condition in vitro meat production system is controlled and manipulatable, it will be feasible to produce design, chemically safe and disease-free meat on a sustainable basis. Despite technical challenges most companies are trying to produce their own versions of lab-growing meat. Even though the production cost, the public acceptance, and huge funds are to a great extent required for further research in the field are the challenge of cultured meat to become commercially feasible.
Keywords
Bioreact; Culture meat
Introduction
World population is rapidly increasing day to day and also meat consumption increases by the same rate. Scientist’s trying to solve this problem by developing lab-growing meat technology from stem cells without animal rearing and slaughter. World Agriculture Towards 2030/2050 Food Agriculture Development Organization (FAO) projections indicate that world population will be increased by more than two billion people from today’s levels, reaching 9.15 billion by 2050 so in vitro meat is an alternative [1]. Worldwide meat production mechanism up-to date uses a number of animals in each year into a food are associated with ethical concerns and are highly criticized for being inhumane [2]. Lab-grown meat is the newest addition to the area called cellular agriculture and many countries and researchers doesn’t give attention up-to-date. Technological advancements like computational simulation highly support cellular agriculture to produce structured products. Cellular agriculture is the mechanism of removing cells from an animal’s tissue and aseptically placing those cells into a controlled environment, and growing those cells.
Meat production from stem cells is the science that secures meat shortage and solves the problem of environmental contamination through animal waste and also protects human health from meat borne disease. Edible animal tissues can be produced by the culturing through tissue engineering and computational simulation techniques from stem cells and grow in the laboratory say that in-vitro meat. In other words cultured (lab-growing/artificial or synthetic) meat [2].
The techniques required to produce lab-growing meat are not over the human practice; this simply involves the culturing of muscle tissue in artificial medium on large scale from farm animal stem cells. Loose myosatellite produced from embryo and muscle biopsy exposed by enzyme and culturing loose myosatellite cells on a substrate, it is one way of to produce cultured meat by harvesting mature muscle cells after differentiation and processing them into various meat products [3]. Starting cells for meat production could be taken from live animals’ biopsy or animal embryos and then put into a culture media where they start to proliferate and grow, independently from the animal. Production of in vitro meat for comminuted and processed meat products, such as sausages, burgers, and nuggets will be easier to develop and the process take place in bioreactor [4].
In vitro meat is an alternative to conventional meat because meat has enjoyed and is still enjoying great popularity, consumers’ concern regarding these issues have risen during the past years [5]. Therefore, the objective this review is:
To provide scientific information about meat production from stem cell, advantages, challenges and prospective.
Stem Cells
Stem cells are unspecialized cells with self-renewal capacity. They can divide through mitosis limitlessly to renew other cell sorts of multicellular living beings all through their life. After stem cell division, each newly created cell can either stay as a stem cell or separate to create any other cell sort with more characterized capacities, such as muscle cell, blood cell, or neural cell. Beneath extraordinary circumstances, separation of stem cells can too be initiated to produce tissue or organ particular cell sorts with extraordinary capacities. There are basically two sorts of stem cells: embryonic stem cells, which are determined from embryos, and substantial or grown-up stem cells, which are undifferentiated cell dwelling in a tissue or organ at the side of other separated cells (Figure 1) [6].
Unique properties of all stem cells
Stem cells differ from other types of cells in the body. All stem cells regardless of their source have three general properties:
1. They are capable of dividing and renewing themselves for long periods.
2. They are unspecialized.
3. They can give rise to specialized cell types.
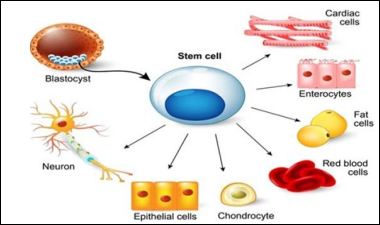
Figure 1: Stem cell differentiation, (source: https://stemcells.nih.gov/info/basics/1. htm).
While differentiating, the cell usually goes through several stages, becoming more specialized at each step. Scientists are just beginning to understand the signals inside and outside cells that trigger each step of the differentiation process. The internal signals are controlled by a cell’s genes, which are interspersed across long strands of DNA, and carry coded instructions for all cellular structures and functions. The external signals for cell differentiation include chemicals secreted by other cells, physical contact with neighboring cells, and certain molecules in the microenvironment. The interaction of signals during differentiation causes the cell’s DNA to acquire epigenetic marks that restrict DNA expression in the cell and can be passed on through cell division [7].
Embryonic stem cells
Embryonic stem cells, as their name suggests, are derived from embryos. Most embryonic stem cells are derived from embryos that develop from eggs that have been fertilized in vitro-in an in vitro fertilization for research purposes with informed consent of the donors (Figure 2).
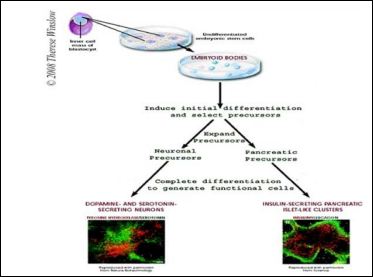
Figure 2: Embryonic stem cell differentiation, (source: https://stemcells.nih.gov/info/ basics/3.htm).
Adult stem cells
An adult stem cell is thought to be an undifferentiated cell, found among differentiated cells in a tissue or organ that can renew itself and can differentiate to yield some or all of the major specialized cell types of the tissue or organ. The primary roles of adult stem cells in a living organism are to maintain and repair the tissue in which they are found [8].
Differences between stem cells and somatic cells
Adult stem cells reside along with somatic cells in many tissues and organs, including peripheral blood, blood vessels, bone marrow, skeletal muscle, teeth, skin, gut, liver, ovary, testis, brain, and heart. They are present in a small number and located in a specific area of each tissue called ‘stem cell niche’. Unlike somatic cells, stem cells can be in an inactive, no dividing state for a long time until they are activated by certain internal or external signals, such as tissue injury or diseased conditions. Adult stem cells can undergo normal differentiation pathways to give rise to specialized cells of the tissue wherein they are located. Some examples of stem cell differentiation into specialized somatic cells are as follows:
Hematopoietic stem cells: Differentiate into all types of blood cells, including Red Blood Cells (RBC), B lymphocytes, T lymphocytes, neutrophils, basophiles, eosinophil’s, monocytes, natural killer cells, and macrophages.
Mesenchymal stem cells: Also known as bone marrow stromal stem cells, differentiate into different cell types, including bone cells, cartilage cells, fat cells, and stromal cells, that regulate blood production.
Neural stem cells: Neural stem cells are present in the brain and can differentiate into three major brain cell types namely neurons (nerve cells), astrocytes, and oligodendrocyte.
Epithelial stem cells: Epithelial stem cells are present in the epithelial lining of the gastrointestinal tract and can differentiate into different cell types, including absorptive cells, goblet cells, and enter endocrine cells.
Skin stem cells: Skin stem cells are of two types: epidermal stem cells that are found in the basal layer of the epidermis and can differentiate into keratinocytes; And follicular stem cells that is found at the base of hair follicles and can differentiate into both follicular cells and keratinocytes.
Normal differentiation pathways of adult stem cells
In a living animal, adult stem cells are available to divide for a long period, when needed, and can give rise to mature cell types that have characteristic shapes and specialized structures and functions of a particular tissue. The following are examples of differentiation pathways of adult stem cells (Figure 3).
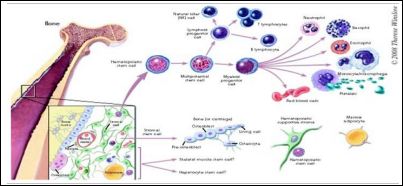
Figure 3: Normal differentiation of adult stem cell, (source:https://stemcells.nih.gov/ info/basics/4.htm).
Stem cell and their function
Stem cells are undifferentiated cells from which other cells originate. Stem cells have the ability either to divide indefinitely or to differentiate into other cell types. Their ability to differentiate varies. Some stem cells differentiate only into cells of certain tissues, while others can differentiate into many cell types. Stem cells are grouped according to their ability to differentiate and their origin. The stem cell that is most able to differentiate is the fertilized ovum. It is the origin of all tissue types and the developing human body. Other stem cells embryonic, fetal and adult stem cells -are much more limited in their ability to differentiate [7].
Stem cells are divided based on source into two categories of embryonic and adult. In another case based on potency categorization stem cells are divided into Totipotent, Multipotent and Unipotent cells. They are vital to the development, growth, maintenance, and repair of our brains, bones, muscles, nerves, blood, skin, and other organs. Stem cells are found in all of us, from the early stages of human development to the end of life. Stem cell research holds tremendous promise for the development of novel therapies for many serious diseases and injuries. Like various blood diseases has been studied (such as lymphoblastic leukemia, myeloid leukemia, thalassemia, multiple myeloma and sickle cell anemia), and other cell therapy in treatment of Parkinson’s disease, Amyotrophic lateral sclerosis, Alzheimer, Stroke, Spinal Cord Injury, Multiple Sclerosis, Radiation Induced Intestinal Injury, Inflammatory Bowel Disease, Liver Disease, Duchenne Muscular Dystrophy, Diabetes, Heart Disease, Bone Disease, Renal Disease, Chronic Wounds, Graft-Versus-Host Disease, Sepsis and Respiratory diseases. The scope of potential stem cell-based therapies has expanded in recent years due to advances in stem cell research [9]. Alopecia areata is one of the most common autoimmune diseases and targets the hair follicles, with high impact on the quality of life and self-esteem of patients due to hair loss. Clinical management and outcomes are challenged by current limited immunosuppressive and immune-modulating regimens [10].
In other than clinical use stem cells are used for meat production using muscle stem cells or mayo satellite stem cells based on tissue culture. Cell culture is a capital venture or vital technique for understanding the structure and function of cells, in recent times it has very good implications in biotechnology. Cultured animal cells are commercially used for the production of interferon, vaccines and clinical materials like growth hormones and also development of viable substitutes that restore the function of damaged tissues and organs [11].
History of Culture Meat
The concept of in-vitro meat for human consumption was ideally written by Churchill before 100 years back and 1932 published in book of thought and adventures. However, it was predicted long back by a writer and Conservative politician Frederick Edwin Smith, 1st Earl of Birkenhead, who predicted that “It will no longer be necessary to go to the extravagant length of rearing a bullock in order to eat its steak. From one “parent” steak of choice tenderness it will be possible to grow as large and as juicy a steak as can be desired” [12].
Willem Van Eelen of the Netherlands independently had the idea of using tissue culture for the generation of in vitro meat in the early 1950s and his idea was to start the new technology of meat production from stem cells. It took until 1999 before van Eelen’s theoretical idea was patented as the concept of stem cells and in vitro culture of cells was yet to emerge.
Harvested muscle biopsies from frogs and kept these tissues alive and growing in culture dishes +and to introduce the new biological tissue culture system [13]. Cultured muscle tissue from the common goldfish (Carassius auratus) in Petri dishes using special media and aiming to explore the possibilities of culturing animal muscle protein for long term space flights or habituation of space stations. A test-panel judged these processed explants and agreed that the product was acceptable as food [14].
As projected by the first in vitro Meat Symposium in 2008 held in as, Norway, the first commercial in vitro meat products will be available in the next 5 to 10 years at prices competitive with European beef (~$5,200-$5,500 per ton or 3,300 to 3,500euros) use in high efficiency of bioreactor [15].
August 2013 in London Professor Mark Post of Maastricht University unveiled the world’s first Cultured Beef burger made from bovine stem cells cocked on Riverside Studios was tested. The burger contained a five-ounce burger patty produced by using laboratory grown beef worth more than $330 000. It took only three months to grow the beef in the laboratory, using stem cells harvested from a cow’s shoulder. He said that according to his work he would expect to see cultured meats in every supermarket in 10 to 20 years the production cost of burger will be 1$ [16].
In the wake of the plummeting legitimacy of factory farming, in vitro meat may very well be a feasible vehicle for both the ethical and pragmatic demobilization of the factory farming industry. Hailed by animal activists and meat experts alike as “victimless meat,” in vitro meat bypasses the moral ramifications of standard meat production, avoiding animal death entirely by typically removing cells from the donor animal via biopsy and cultivating cells in medium containing mushroom extract rather than animal blood serum [17].
Mechanism of production
Tissue Engineering is a pioneering field and bioreactors have an important role in creating the ideal environment for the generation of a particular new tissue. Requirements of in vitro meat production are Firstly; a cell source is required that can proliferate indefinitely and also differentiate into functional skeletal muscle tissue. Secondly, these cells need to be embedded in a three-dimensional matrix that allows for muscle growth, while keeping the delivery of nutrients and release of waste products undisturbed and lastly, muscle cells need to be conditioned adequately in a bioreactor to get mature, functional muscle fibers for processing to various meat products. And For the growth of any substance, an affordable medium is required. Such complete nutrients must be supplied because the digestive system is not available. Deserve dignity for Benjaminson and others succeeded in using a serum-free medium made from mushroom extract that achieved higher rates of growth than fetal bovine serum [14].
Cell source
The best sources of stem cells are adult and embryonic stem cells. Stem cells from embryos can be isolated from 5-7-day-old blastocysts. Adult stem cells can be found in the human body, but sufficient quantities of adult stem cells for therapeutic purposes can only be isolated from bone marrow. But for the meat production purpose is muscle cell or my satellite stem cell. An adult stem cell is an undifferentiated cell found among differentiated cells in a tissue or organ, can renew itself and can differentiate to yield the major specialized cell types of the tissue or organ. The primary roles of adult stem cells in a living organism are to maintain and repair the tissue in which they are found [18].
During culture meat production satellite cell isolation is the first work. The methodology employed to isolate and culture satellite cells in vitro can critically determine the fusion of myoblasts into multinucleated myotubes. Hence, a simple method of muscle satellite cell isolation and culture of different vertebrate species that can result in a high fusion rate is highly desirable and the cell culture conditions, excellent fusion rates were achieved in the duck, chicken, horse and cow (with more than 50% cell fusion) [19].
Three-dimensional matrix
It is the hard material in micro or Nano level used for supporting the differentiation of stem cells into muscle fiber and also to produce the wanted product. Matrices can be divided into randomly orientated scaffolds and matrices with a certain alignment. Commonly used matrices with random orientation, like gels and sponges, can be used for a variety of tissues. However, in case of skeletal muscle this tissue naturally consists of bundles of highly oriented muscle fibers in an extracellular 3D matrix to form an organized tissue with high cell density [20].
Bioreactor
Bioreactors may be in vivo or in vitro based on the processes taking place. In vitro bio-reactors are used like: Static Culture bioreactors have been widely used in the past and involve the deposition of cells on a scaffold, supplied with the appropriate growth media, and cultured in an incubator. Rotating Wall Vessel bioreactor was developed at NASA (National Aeronautics and Space Administration) and consists of two concentric cylinders, within which lies an annular space containing the scaffold with dynamic laminar air flow. Spinner Flask in this type, cell seeded scaffolds attached to needles are suspended from the top cover of a flask in culture medium. It seems to support ontogenesis more than the rotating wall vessel bioreactor. Flow Perfusion bioreactors utilize a pump to percolate medium continuously through the scaffolds interconnected pores and eliminate the internal transport limitations of the spinner flask and rotating wall vessel [21].
The process of developing a cultured-meat production system involves the initial collection of suitable cells with the potential to form muscle, as outlined in the previous section, and then greatly expanding the numbers in a bioreactor [22]. Strips of muscle fiber can be generated, but the development of a full-scale bioreactor suitable for mass cultured meat production has yet to be designed and built [3].
Skeletal muscles are composed of bundles of highly oriented and dense muscle fibers, each a multinucleated cell derived from myoblasts (Figure 4). The muscle fibers in native skeletal muscle are closely packed together in an extracellular three-dimensional matrix to form an organized tissue with high cell density and cellular orientation to generate longitudinal contraction [11].
Self-organizing / Tissue culture techniques
This technique is the first technique and was employed by Benjaminson, Gilchriest and Lorenz they practice on the fish muscle development explants from goldfish (Carassius auratus) in diverse culture media for seven days and observed an increase in surface area in different mediums was different percentage. The results-based on the medium was as fetal bovine serum: 13.8 %, fishmeal extract: 7.1 %, shiitake extract: 4.8 %, maitake extract: 15.6 %. The explants were also placed in a culture containing dissociated Carassius skeletal muscle cells and an increase of 79 % in the explants surface area was recorded [14].
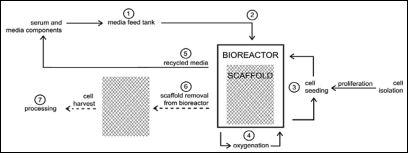
Figure 4: Bioreactor design the different design approaches for an in vitro meat production system can be roughly divided into scaffold/cell culture based and self-organizing/tissue culture techniques.
Self-organizing in vitro meat production may hold the promise to produce the highly structured meats as the explants contain all the tissues which make up meat in the right proportions and seems like in vivo situation. The Self organizing technique helps to create structured meat i.e. meat produced will have a well-defined 3-D structure, just as the natural conformation of meat. The same can be achieved using the principles of tissue engineering for de novo synthesis of muscle tissue [5].
Cell culture /scaffold-based technique
It cannot produce highly structured meats like steaks but can be used to produce ground and boneless meats with soft consistency. Scaffolds are porous biomaterials and play a pivotal role in the Tissue Engineering paradigm by providing temporary structural support, guiding cells to grow, assisting the transport of essential nutrients and waste products, and facilitating the formation of functional tissues and organs. This was the second type of meat production technique. The principle of scaffold-based technique is that suitable muscle cells proliferate on a carrier called scaffold in the presence of a culture medium in a bioreactor. Technicians would isolate embryonic or adult stem cells from a pig, cow, chicken or other animal. Then they would grow those cells in bioreactors, using a culture derived from plants. The stem cells would divide and re-divide for months on end. Technicians would next instruct the cells to differentiate into muscle (rather than, say, bone or brain cells). Finally, the muscle cells would need to be “bulked up” in a fashion similar to the way in which animals build their strength by exercising. Scaffolds developed by using natural and edible biomaterials like collagen that allow for 3-D tissue culture and complex structuring of meat have also been proposed and attempted [17,23].
Advantages of Culture Meat
The main important advantage of producing cultured meat is better control over meat composition and quality by manipulating the composition of the culture medium or co-culturing with other cell types like flavor, fatty acid composition, fat content and ratio of saturated to poly-unsaturated fatty acids. Harmful saturated fats could be replaced by healthy fats, like omega-3. In other hand to enhance human health add certain vitamins into culture media [24].
The conventional meat production practices have some risks like zoonotic disease, shortage of grassland, drug resistance disease in the environment and also greenhouse gas emission is increasing etc. but the in vitro meat production system is that the conditions are controlled and manipulated to reduce the risk [3]. It will be easier to keep control of pathogenic contamination in cultured meat production, but in vitro meat is associated with more risks of contamination of substrates and fewer risks with respect to microbial contamination [25].
Culture meat is an all-embracing feeding style. Vegetarians eat some animal product like egg, milk, white chicken and fish but they prohibit meat from cattle, sheep, goat and the like. This becomes particularly interesting when we bring cultured meat into the frame. In the production of meat foodstuffs in this category, there is no animal suffering; there is no exploitation, no slaughter, merely the growth of cells in a fermentation vat or tank. Even a vegetarian could, in that case, in principle consume cultured meat [12].
Environmental Implications of cultured meat in comparison to conventionally produced meat, it involves approximately 7_45% lower energy use (only poultry has lower energy use), 87% lower greenhouse gas emissions, 99% lower land use, and 89% lower water use depending on the product compared. Despite high uncertainty, it is concluded that the overall environmental impacts of cultured meat production are substantially lower than those of conventionally produced meat [26].
The negative impact of conventional meat production is environmental disturbance and shrinkage of resource (Figure 5) [27].

Figure 5: Environmental disturbance.
In the case of animal welfare in vitro meat production is positively implication and acceptable by animal activists and meat experts use as “victimless meat”, in vitro meat bypasses the moral ramifications of standard meat production, avoiding animal death entirely by typically removing cells from the donor animal via biopsy and cultivating cells in medium containing mushroom extract rather than animal blood serum [15,17]. In theory a single farm animal may produce and supply the world population. If ten stem cells divide and differentiate continually for two months, they could yield 50000 metric tons of meat [23]. Farther more the dramatic reduction in land use opens the prospect that much of this land may be used for other purposes or just returned to wilderness which may help in restoration of many endangered species [28].
Culture meat is to implication for space mission or travel settlements current trend, supply and physicochemical regeneration (of water and oxygen) are the most cost-effective, but for longer periods and permanent bases, bio-regeneration becomes more attractive [28]. A controlled ecological life support system (cells) would not only provide fresh food to the astronauts, but also deal with waste, and provide oxygen and water [14,28,29]. Due to Good Manufacturing Practices, the incidence of food borne diseases could be significantly reduced as the chance of meat contamination would be lower in absence of a potentially compromised organism. In addition, the risks of exposure to other hazards associated with conventional meat production systems like pesticides, arsenic, dioxins, and hormones could be significantly reduced [27].
Disadvantage of Culture Meat
Edelman et al. already described in 2005 that the techniques that are necessary for the production of in vitro meat were not beyond imagination. It is also stated however, that these techniques are only able to produce boneless meat like hamburger or sausages. But the production of higher structured meat like steak is too complex. In vitro meat production is theoretically possible, but it faces greater technical as well as financial challenges is the major problem. In addition to the fund’s benefit of culture, meat from stem cells has the major challenge is color and appearance is difficulties in competing with the conventional meat. Mark post in London to test the culture meat was reported it was colorless. The color of the meat was improved by adding a bit of red beet juice and saffron [17].
Unnaturalness of cultured meat and source of animal is being perceived as one of the strongest barriers for public acceptance like halal in Islam, Jewish and vegans [30]. People may feel in vitro meat as artificial meat and not the real thing and as such they depreciate the value of the meat in the same way they would look down on artificial flowers or synthetic diamonds [17].
As a prospect in the Christian follower the lord orders on the holy bible Deuteronomy chapter 14:3-21 to list out the name of edible animal meat and not touchable animals and also affect the source of stem cells. ” thou shalt not eat any abominable thing the best you shall eat ox, sheep and goat’’. And nevertheless, you shall not eat camel and swine. It also shares with Jewish.
The first “In vitro Meat Symposium” in 2008, held in Norway, indicated that the first commercial in vitro meat products would be commercially available in the next 5 to 10 years at prices competitive with European beef (~$5,200–$5,500 per ton or 3,300-3,500 and each individual can access easily in the supermarket [31]. However, in vitro meat production at a commercial level still requires significant in-depth research because In the near future, cultured meat will be an essential part of human diet; nonetheless, in the short term, the extremely high cost of biosynthesized meat is the main hurdle to its feasible commercialization so researchers digging hardly/devote their knowledge and time/ to improve the production cost minimization and commercialization [32].
A great challenge in producing a competitive in vitro grown meat product is ensuring that all necessary nutrients are present. Dietary minerals and vitamins not synthesized by myocytes will often require binding proteins in medium and effective transport mechanisms for entry into the cells. Knowledge of the complex metabolism of each crucial vitamin and mineral is necessary to develop a nutritionally valuable meat product. While determining the proper nutrient profile will be a major hurdle to overcome, it comes with the knowledge of how to manipulate the culturing system to make nutritionally tailored products [3].
Future Prospects
Feasibility of in-vitro meat production will be solving many problems related to conventional meat production. But these processes required developing bioreactors and to investigate the way that they mimic natural meat by all content (structure morphological and nutritional value). In addition to this the production cost, the public acceptance, and huge funds are to a great extent required for further research in the field are the challenge of cultured meat becomes commercially feasible.
Conclusion
World population is rapidly increasing day to day and also meat consumption increases by the same rate. Conventional meat production systems require a relatively high proportion of land, energy, and water use. Nutritional related disease food borne illnesses, development of antibiotic-resistant pathogen strains, and animal welfare issues are the other factors. Production of in vitro meat by culturing muscle cells of farm animal species with the aid of computational simulation technology seems to be one of the prospective solutions to get structured meat. Besides reducing the use of animals, it may combine a favorable ecological footprint with similar nutritional values and sensory qualities as that of the conventional meat. In-vitro meat production will be held great, promising to provide consumer resistance overcome and a great deal will be performed with respect to the cost effectiveness of the technology, and ethical and societal issues before effective large-scale production can be achieved.
References
- United Nations World Population Prospects, the 2012, Revision, 3-6.
- Kadim IT, Mahgoub O, Baqir S, Faye B, Purchase R (2015) Cultured Meat From Muscle Stem Cells: A Review of Challenges and Journal of Integrative Agriculture 14: 222-233.
- Bhat ZF, Bhat H (2011) Tissue Engineered Meat- Future Meat. Journal of Stored Products and Postharvest Research 2: 1-10.
- Datar I, Betti M (2010) Possibilities for an in vitro Meat Production Innovative Food Science and Emerging Technologies 11: 13-22.
- Edelman PD, McFarland DC, Mironov MD, Matheny JG (2005) In vitro-Cultured Meat Production (Commentary). Tissue Engineering 11: 659-662.
- Roelen BA, Chuva de Sousa Lopes SM (2008) Of Stem Cells and Gametes: Similarities and Differences. Current Medicinal Chem- istry 15: 1249-1256.
- Yin H, Price F, Rudnicki MA (2013) Satellite Cells and the Muscle Stem Cell Niche. Physiological Reviews 93: 23-67.
- Slack JM (2008) Origin of Stem Cells in Organogenesis. Science 322: 501-1498.
- Kalra K, Tomar PC (2014) Stem Cell: Basics Classification and American Journal of Phytomedicine and Clinical Therapeutics 2: 913-930.
- Li Y, Yan B, Wang H, Li H, Li Q, et al. (2015) Hair Regrowth in Alopecia Areata Patients Following Stem Cell Educator BMC Medicine 13: 1-11.
- Bach AD, Beier JP, Stern-Staeter J, Horch RE (2004) Skeletal muscle tissue engineering. J Cell Mol Med 8: 524-540.
- Ford BJ (2011) Culturing Meat for the Future: Anti-Death versus Anti-Life. 1-26.
- Catts O, Zurr I (2011) The Tissue Culture Sculptures: Art project. Leonardo 35: 365-370.
- Benjaminson MA, Gilchriest JA, Lorenz M (2002) In Vitro Edible Muscle Protein Production System: Stage 1 Fish. Acta Astronau- tica 51: 879-889.
- Alexander R (2011) In Vitro Meat: A Vehicle for the Ethical Rescaling of the Factory Farming Industry and In Vivo Testing or an Intractable Intersect: The Stanford Journal of Science 4: 42-47.
- Zaraska M (2013) Lab-Grown Beef Taste Test ‘Almost’ Like a Health & Science.
- Hopkins PD, Dacey A (2008) Vegetarian Meat: Could Technolo- gy Save Animals and Satisfy Meat Eaters? Journal of Agricultural and Environmental Ethics 21: 579-596.
- Kumar S, Singh N (2006) Stem Cells New Indian Journal of Human Genetics 12: 4.
- Baquero-Perez B, Kuchipudi SV, Nelli RK, Chang KC (2012) A Simplified but Robust Method for the Isolation of Avian and Mammalian Muscle Satellite Cells. BMC Cell Biology 13: 2-11.
- Klumpp D, Horch RE, Kneser U, Beier JP (2010) Engineering Skeletal Muscle Tissue New Perspectives in Vitro and in Vivo. Journal of Cellular and Molecular Medicine 14: 2622-2629.
- Oragui E, Nannaparaju M, Khan WS (2011) The Role of Bioreac- tors in Tissue Engineering for Musculoskeletal Applications. The Open Orthopaedics Journal 5: 267-270.
- Post MJ (2012) Cultured meat from stem cells: Challenges and Prospects. Meat Science 92: 297-301.
- Bartholet J (2015) Technology inside the Meat Lab. Scientific American 304: 106-111.
- Christine Mummery, Willem Frederik Van Eelen, Willem Jan Van Kooten, Wiete Westerhof (1999) Industrial Scale Production of Meat from in vitro Cell Culture. International Search Report.
- Welin S (2013) Introducing the New Meat Problems And Prospects. World’s Research 7: 24-37.
- Tuomisto, Teixeira de Mattos (2011) Environmental impacts of cultured meat Environmental Science and Technology 45: 6117-6123.
- Bell AW, Charmle E, Hunter RA, Archer JA (2011) The Austral asian beef industries Challenges and opportunities in the 21st Animal Frontiers 1: 10-19.
- Bhat ZF, Kumar S, Fayaz H (2014) In vitro meat production: Challenges and benefits over conventional meat Journal of Integrative Agriculture 14: 241-248.
- Drysdale AE, Ewen MK, Hanford AJ (2003) Life Support Approaches For Mars Missions. Advances in Space Research 31: 51-61.
- Saha PR, Trumbo PR (1996) The nutritional adequacy of a limited vegan diet for a controlled ecological life-support Advances in Space Research 18: 63-72.
- Jennifer P (2018) Cultured Meat: Lab-Grown Beef and Regulating the Future Meat UCLA Journal of Environmental Law and Policy 36: 104-126.
- Morton JD, Mason SL, Bekhit AEA, Bhat HF, Bhat ZF (2019) Technological, Regulatory, and Ethical Aspects of In vitro Meat : A Future Slaughter-Free Harvest. Wiley Online Library 18: 1192-1208.
Citation: Mengistie D (2020) Lab-Growing Meat Production from Stem Cell. J Nutr Food Sci 3: 015.
Copyright: © 2020 Mengistie D. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and re- production in any medium, provided the original author and source are credited.


