*Corresponding Author:
Massimo Milani,
Medical Department Cantabria Labs Difa Cooper, Italy
E-mail: massimo.milani@difacooper.com
Abstract
Emu Oil (EO) is a product derived from the Emu (Dromaius novaehollandiae). The Emu is a large wingless bird whose oil was used by Australian aboriginals to accelerate wound healing and alleviate local pain. EO has been demonstrated to have significant anti-inflammatory effects. When used topically, EO is also an effective emollient and skin barrier protecting substance. The principal characteristic of EO is its peculiar composition of long chained triglyceride esters (FFA) of omega (Ω) 3, 6 and 9 classes, such as Oleic and Linoleic acids and also saturated fatty acids like palmitic and stearic acids. EO also contains other components such as carotenoids, flavonoids, polyphenols, and tocopherols, which can result in favourable antioxidant effects of this product. The FFA of the Ω-3, Ω-6, and Ω-3 series, which are the main component of EO, may act on cyclooxygenase, lipoxygenase, and lipoxin pathways expressing anti-inflammatory actions. More recent investigations support the potent and specific anti-inflammatory effects of EO when used both orally and topically. EO in fact can induce a downregulation of IL-1, IL-6 and TNF-α cytokines production in in vitro and in vivo experiments. It has been suggested that EO can act as a natural precursor to the body’s own anti-inflammatory agents, working in different ways to relieve inflammation processes. Topical formulations containing high concentration of EO have demonstrated to be effective in improving clinical signs and symptoms (i.e., erythema and itch) in subjects with subacute atopic dermatitis and seborrheic dermatitis manifestations. EO could also improve the skin barrier function. EO stimulates skin cells proliferation, favouring wound healing process. This review focuses the available evidence of EO use in dermatology and the potential mechanisms of action.
Keywords
Eczema; Emu oil; Omega 3-6-9 free fatty acids; Wound healing
Introduction
The Emu Oil (EO) is extracted from the subcutaneous and retroperitoneal fat of Emu, a flightless bird living in Australia (Figure 1) but now it is also farmed in Canada, Europe and the USA [1]. This product is rich in free fatty acids and was used in traditional medicine for treating wounds and pain reduction for inflamed joints [2]. EO is available in both topical and oral administration formulations [3]. The principal characteristic of EO is its peculiar composition of long chained triglyceride esters (FFA) of omega (Ω) 3, 6 and 9 classes, such as Oleic acid and Linoleic acid and saturated fatty acids like Palmitic acid and Stearic acid [4]. EO, from a pharmacological point of view, is characterized by antioxidant, anti-inflammatory and skin reparative activities [5]. EO is mainly used topically but recent trials have shown that EO could exert anti-inflammatory action also after oral administration [6]. This review focuses the available evidence of topical formulations of EO regarding the potential mechanisms of actions and the use in dermatology. In this review we summarized the available published data (up to 2021) regarding the role of emu oil in dermatology with a particular focus in term of composition, mechanism of action, efficacy data and future challenges. We performed a Pubmed search (September 2021) to identify and extract information regarding Emu oil in dermatology using the following terms: emu oil AND skin AND/OR clinical trial AND/OR topical formulation. In comparison with similar reviews on the same topic (Mashtoub 2017), we found more recent articles providing additional new data on mechanism of action (in particular new anti-inflammatory mechanisms and stem cell promoting activity of emu oil) and new clinical efficacy data in dermatology conditions such as atopic eczema and skin barrier function.

Figure 1: Emu (Dromaius novaehollandiae) is a wingless bird living in Australia. The Emu Oil (EO) is extracted from the subcutaneous and retroperitoneal fat of this ani- mal (source Wikipedia; open source image).
Chemical Composition of EO
The EO is produced though extraction, filtration, and centrifugation processes from the subcutaneous and retroperitoneal fat of the animal [7]. When the semi-solid fat is filtered at 25 °C it yields 2080% (v/v) of a clear bright yellow oil. The final product could be used for the set-up of oral and topical formulations. EO is commonly approved by regulatory Agencies, such as FDA. EO contains 98% of lipid substances. The predominant component of EO is represented by a peculiar mix of unsaturated (UFA) and saturated (SFA) Free Fatty Acids (FFA) such as Oleic, Linoleic, Palmitic and Linolenic acids. More in detail, the EO composition is formed by 45% of monounsatured, 35% of satured and 20% of polyunsatured FFA. Table 1 describes the qualitative-quantitative composition of EO. The content of α-linolenic acid in the total triglyceride fraction could vary according to farming conditions and the basal diet. The Figure 2 shows the percentages of monounsaturated, saturated and polyunsaturated fatty acids present in standardized EO formulations. The relevant point is that the FFA qualitative and quantitative composition of EO is very similar to the FFA composition of human skin [7]. In addition to this lipid component, EO contains also natural antioxidant molecules such as flavonoids, carotenoids, tocopherols, polyphenols and finally phospholipids. When EO is used to produce topical formulations the issue of standardization of the composition could be a relevant critical aspect. The EO composition in fact can depend upon the age of the bird, the type of diet and of course the extraction and production methods. In addition, there are no standardized procedures for the EO production. Anyway, a trial [8] conducted with different EO products has shown similar pharmacological effects in a model of mucositis, suggesting that different EO commercially available products do not substantially differ in term of pharmacological activity and clinical efficacy. One relevant advantage of EO is that its production requires little processing, unlike plant derived oils, and presents a high safety and tolerability profile, being readily metabolized like most animal fats. Furthermore, in contrast to so many petroleum-derived pharmaceuticals, it also comes from a eco-sustainable and renewable resource. It is relevant to note that EO is nonirritating, possessing good moisturizing and cosmetic properties with low comedogenicity and good penetrating ability across stratum corneum compared with other mineral oils. Therefore, EO is of major interest to dermatologists and cosmetic scientists as an interesting compound for topical formulations.
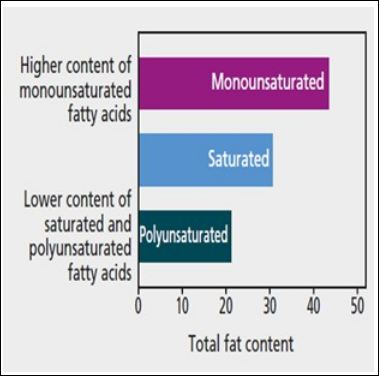
Table 1: Qualitative and quantitative composition of EO*.
** SFFA: Satured free fatty acid. *Source: Mashtoub 2017
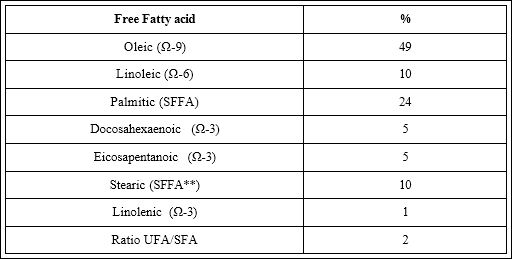
Figure 2: EO: Percentages of monounsaturated, saturated and polyunsaturated fatty acids. Total fat content in % of weight.
Pharmacological Activities of EO
The EO has several relevant pharmacological activities demonstrated by several in vitro and in vivo studies. Scientific evidence supports the data that EO has antioxidant, anti-inflammatory and skin barrier reparative functions.
Antioxidant activity
The antioxidant action of EO was demonstrated both in vitro and ex vivo models [9]. EO has a scavenging property, and it is also able to inhibit lipid peroxidation in a biological membrane model. Bennet et al [10] demonstrated that the antioxidant activity of EO was superior to other avian oils. These authors stated that the antioxidant or radical scavenging properties of EO appeared to be due to minor constituents in the non-triglyceride fraction of the product, while its high ratio of unsaturated to saturated fatty acids (UFA/SFA>2) could offer protection against oxidative damage.
Anti-inflammatory activity
The anti-inflammatory action of EO was demonstrated in several animal models [11,12]. EO applied topically significantly decreases auricular thickness by more than 70% in a mouse model of croton oil-induced auricular inflammation. When used orally, EO can counteract inflammation processes in animal models of acute intestinal mucosa inflammation [13]. Interestingly, EO reduces the production of pro-inflammatory cytokines such as tumor necrosis factor and interleukin 1-α [14]. The anti-inflammatory action of EO could be due to the high content of PUFA present in the EO like docosahexaenoic and eicosapentanoic acids [15]. In addition, PUFA can reduce the production of IL-12 and IL-1β [16]. Recent data show that EO emu oil might regulate PPARγ and TNFa [17] expressining a relevant anti-inflammatory action Therefore, the presence of Ω-3 and Ω-9 FFA in the EO could explain the anti-inflammatory activity of this product with both a direct and indirect mechanisms of action (reduction in leukot-riene and thromboxane production, down-regulation in inflammatory genes expression). Finally, the Ω-9 FFA component of EO can inhibit macrophage migration [18]. Thus, the anti-inflammatory action of EO could be considered to act at multiple levels Figure 3.

Figure 3: Proposed mechanisms of action of Emu oil.
Skin Barrier reparative functions and regeneration of skin stem cells
EO has been proven to stimulate the proliferation of the skin. In 2018 Arenzoumand et al [19] demonstrated, in an in vitro model, that an emulsion of EO enhanced the proliferation, stemness gene expression and the wound healing capacity of adipose-derived stem cells. Furthermore, Lopez et al [20] demonstrated that EO could promote wound healing in experimentally induced lesions in mice. EO has relevant moisturizing effects: Zanardo et al [21] have shown that a single topical application of an EO-based lotion in newborns was effective in improving stratum corneum hydration and skin elasticity. Skin hydration increased by 22% just 1 hour after the application of the EO-based lotion and by 34% after 24 hours. The same research group [22] demonstrated that topical application of EO-based cream improved the skin barrier function of the areola skin in breastfeeding women. EO topical use can have interesting wound healing promoting activity.
Pharmacology of Omega 3, 6 and 9 FFA
In consideration of the fact that EO is a very high rich mixture of Ω-3, Ω-6 and Ω-9 FFA, a general description of pharmacological main activities of these biological compounds is relevant to understand the utility of EO in clinical use. FFA are important energy sources for most body tissues and are classified according to their aliphatic tail length; Short-Chain Fatty Acids (SCFAs) have fewer than 6 carbon atoms, Medium-Chain Fatty Acids (MCFAs) have 6-12 carbons, and Long-Chain Fatty Acids (LCFAs) have 12 or more carbons atoms. Unsatured and Polyunsaturated Fatty Acids (PUFAs) are fatty acids that contain one or more than one double bond in their backbone. This class includes many important compounds, such as essential fatty acids. In more details, FFA are formed by a chain of hydrocarbons with a carboxyl group (-COOH) group at one end, and a methyl group (-CH3) at the opposite side. The carbon next to the -COOH is known as α, the next one β. Therefore, the last position is labelled as a “ω”, the last letter in the Greek alphabet. The Ω-3 or 6 or 9 classification refers on the position of the first unsaturation bond relative to the end position (ω). For example, the term ω-3 means that the first unsaturated carbon-carbon bond from the terminal end (ω) of then chain is the third one. Typically, the number of carbons and the number of double bonds is also listed in short descriptions of unsaturated fatty acids. From a strict pharmacological point of view, FFA are relevant molecules showing critical functions such as receptor signaling, gene expression, and regulation of systemic metabolic energy homeostasis. At intracellular level there are specific physiological sensors for FFAs which are members of the intracellular or nuclear lipid-binding protein families, such as Fatty Acid Binding Proteins (FABPs) and Peroxisome Proliferator Activated Receptors (PPARs), are known as functional receptors that regulate many physiological and pathophysiological processes stimulated by FFA. In addition, there are specific membrane receptors of the G-coupled family which could be activated by Ω-FFA [23]. For example, the membrane receptors GPR40 and GPR120 are activated by FFA (more specifically by Ω-3 FFA). The activation of these receptors, in particular the FFAR1 receptor, can start anti-inflammatory response blocking the production of one of the key pro-inflammatory molecules such as NF-κ-Beta [24]. Fujita et al. [25] demonstrated that FFAR1 activation blocks the production of cytokines and chemokines via proinflammatory cytokines in keratinocytes and lessens allergic inflammation in the skin. Moreover, a recent Nagatake et al. [26] have shown that some FFA can act as a FFAR1 ligand and exhibits antiallergic and anti-inflammatory effects by inhibiting neutrophil mobility in animal models of contact hypersensitivity.
Topical EO Use in Dermatology
Fatty acids are relevant components of natural lipids, which regulate the physiological structure and function of the human skin. EO, as a source of FFA, can attenuate skin irritation and inflammation and this is the reason why it is used in the treatment of numerous skin diseases. EO in topical formulations has been used in clinical dermatological conditions such as atopic dermatitis, seborrheic dermatitis, and radiation dermatitis and in wound healing. There are also experimental data supporting a significant role of EO in reducing the scar formation after burn.
Atopic Dermatitis and Eczema
Atopic Dermatitis (AD) is a common chronic inflammatory and eczematous skin condition characterized by flares and remissions [27]. AD affects approximately 20% of children in industrialized countries. Several data support the concept that skin barrier alteration or dysfunction and the consequent cutaneous inflammation are the two main hallmarks of AD and they are considered the most relevant pathogenetic factors in this skin disease [28]. The downregulation of cornified envelope genes such as the protein filaggrin, the reduction of ceramide levels, the increased levels of endogenous proteolytic enzymes, and the enhanced trans-epidermal water loss seem the relevant pathogenetic factors responsible of the defective epidermal barrier function [29]. An increase of trans epidermal water loss also in uninvolved AD area is a characteristics of AD skin. The alteration of skin barrier function in AD promotes the local production of primary proinflammatory cytokines such as interleukin 1 (IL-1) and tumor necrosis factor alfa (TNFα). Topical corticosteroids are the mainstay treatment of AD, especially during flare periods however, their use in the long term could cause alteration in skin barrier function. For these reasons, the daily use of emollients and moisturizers is considered an important adjunctive strategy to improve skin barrier function and skin appearance in AD patients [30]. A functionally normal epidermal barrier function is crucial in preventing excessive water loss and protecting skin from external insults such as bacterial, allergen, chemical and mechanical damages. Skin hydration is now considered a relevant component of the overall management of patients with atopic derma-titis. Lotions, which have a high water and low oil content, can worsen xerosis via evaporation and trigger a flare of the disease. For this reason, emollient formulations rich in fat are preferred in this condition. The epidermis in AD is characterized by a significant decrease in the lipid content when compared to the healthy controls [31]. In an animal model of AD dietary supplementation of omega-6 and omega-3 essential fatty acids improved the skin barrier function [32]. Interestingly, a study in an animal model conducted by Son et al [33] demonstrated that the Free fatty acid receptor 4 (FFA4) activation can ameliorate 2,4-dinitrochlorobenzene-induced atopic dermatitis by increasing regulatory T cells function in mice. These data represent a strong rational for the use of EO in AD. Two topical formulations of EO (a lipogel with 90% of EO and a cream containing 25% of active principles) have demonstrated [34,35] to improve the clinical signs of atopic dermatitis in 44 atopic children (mean age 9 years) when used in acute phase (the lipogel formula) and in the chronic phase (the cream product). After 4 weeks of treatment (twice daily application) with the lipogel in the acute phase followed by 8 weeks of treatment with the cream in the post-acute phase 70% of the subjects improved significantly with a reduction of the Investigator Global Assessment (IGA) score greater than 50%.
Seborrheic Dermatitis
Seborrheic Dermatitis (SD) is a common chronic-recurrent skin disease which could affect up to 15% of adult population. SD is more common in men between the age of 20 and 45 years. Skin areas rich in sebaceous glands, such as face, and chest are commonly affected [36]. A key pathogenetic role in SD seems to be played by Malassenzia yeasts which could be responsible to alter the skin barrier function and to start inflammatory response [37]. In a 3-arm, split-face, randomized study [38] conducted in 126 subjects affected by Seborrheic Dermatitis (SD) EO was compared to clotrimazole and hydrocortisone. EO was superior to clotrimazole in reducing the erythema score but inferior to the corticosteroid. However, EO treatment reduced significantly, in comparison with baseline, scaling and itch. The authors stated that EO could be considered as a potentially useful agent that significantly improves itching, erythema and scales associated with SD.
Radiodermitis and Mucositis
Radiation therapy is used in the treatment of many patients with cancer. The utilization of radiation therapy is sometimes limited by the occurrence of radiation-induced skin changes. In fact, skin reactions are common side effects of radiation therapy, with up to 90% of patients being affected. Radiation-induced skin changes are classified into acute and chronic, with acute erythema and desquamation as well as chronic atrophy and fibrosis. So far, the prevention and management of radiation-induced skin injury remains a challenge. EO was evaluated in a placebo-controlled trial [39] in 45 subjects undergoing a cycle of radiotherapy with the aim to evaluate the potential effect of EO in preventing radiation dermatitis. In comparison with placebo EO reduced the global skin damage score however the clinical effect was evaluated as modest-moderate. EO has also shown [40] promising results in subjects with burned wounds in order to reduce the risk of scarring formation: in comparison with placebo EO improved significantly the pliability and pigmentation of the affects skin.
Skin Ageing
Skin ageing is defined as an enhanced process of degradation of epidermal and dermal structural integrity and functionality as a con- sequence of exposition following to many environmental factors such as tobacco smoke, exposure to Ultra-Violet Radiation (UVR) and air pollution [41]. Skin aging may influence epidermal lipids and free fatty acid composition, and their physiological functions may be im- plicated in aging process. A decrease of FFA content in epidermis and dermis has been demonstrated in advanced photoageing [42]. Some FFA can inhibit the activity of Matrix-Metallo Proteases (MMP), a family of enzymes involved in the skin ageing processes: the study of Nicolai et al. in fact demonstrated that omega-3 and omega-6 fatty ac- ids inhibit the proteolytic activity of MMP-2 and MMP-9 [43]. These data are supported also by the study of Rogert et al [44] demonstrating a significantly decreased levels of all major lipid species, in particular ceramides, with increasing age. The FFA of the Ω-3 family are consid- ered important molecules with photoprotective action [45], suggest- ing that this FFA component can counteract the photoageing process. More recently Kazem Nejati-Koshki [46] demonstrated that EO en- hances cell proliferation preserving stem cells function in an in vitro model of cultured adipocytes. High levels of Ω-3 Ω-6 FFA can also be relevant in fighting the skin damage induced by pollution [47]. This amount of evidence, represents a potential rationale implying that EO could be a component with anti-aging action, suggesting a role of EO- based products also in dermo-cosmetic treatment protocols.
There are in addition several practical considerations which fur- ther support the interesting role of EO in dermatology. It has been demonstrated that EO has an excellent safety and tolerability profile: it has no side effects and that, at its maximum dosage, it has a very low skin irritation potential. Another important property of EO is the lack of comedogenic activity, that is, it does not block the skin pores and therefore does not cause the onset of acne in the skin areas where it is applied. In 1996 Zemtsov [48] demonstrated in a placebo-controlled study that EO topical product has high cosmetic and moisturizing properties. EO could therefore be an interesting adjunctive compo- nent for specific anti-inflammatory and anti-acne topical products. Ta- ble 2 reports a list of potential use of EO in dermatology and the type of experimental data available.
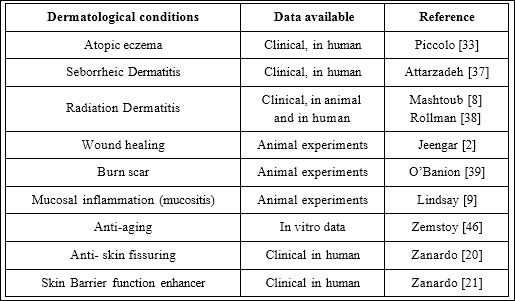
Table 2: EO topical use in dermatology.
Discussion
Emu oil contains high amount of polyunsatured fatty acids and antioxidants. The composition of FFA is quite similar to the lipid mix of human skin. EO has shown to be a potent anti-inflammatory compound and this action could be exerted both orally and topically. EO applied topically has also a stem cell stimulating activity. EO has also a transdermal penetration enhancing effect. The presence of Ω-3, Ω-6 and Ω-9 FFA in EO can explain at least in part the anti-inflammatory action of this product acting on cyclooxygenase and lipoxygenase pathways. For example, Ω-3 FFA can stimulate a specific cell receptor (such as GPR40, GPR120, FFRA1 ect.) and this activation interferes with the pro-inflammatory cascade process mediated by the activation of TLR-4 with the stimulation of NF-κB, a key factor in inflammation response. EO in topical formulation has shown to be beneficial in clinical condition like atopic dermatitis, seborrheic dermatitis, wound healing and in the prevention of radiation dermatitis. EO topical use could enhance the skin barrier function in newborn, therefore representing an interesting strategy in clinical conditions characterized by an altered skin function such as xerosis of atopic skin or xerosis of diabetic skin. In addition, EO could be potentially useful in anti-aging and anti-acne products.
Conclusions
EO, thanks to its peculiar composition, very rich in free fatty ac- ids, and mechanism of action, could be considered as an attractive pharmacological agent. When used topically EO increases the skin barrier function, has antioxidant, anti-inflammatory and emollient ef- fects and it could be used as relevant coadjuvant treatment in several skin conditions. Clinical data suggest that the topical use of emu oil could exert beneficial effect in a wide range of dermatological con- ditions. However, we need more evidence-based robust data on the efficacy and tolerability profile of this interesting product. Therefore, further trials with comparative or add-on study design are warranted to better define the role of topical emu oil in dermatology. Finally, a relevant challenge to better describe the role of topical emu oil would be the issue of standardization of pharmaceutical preparations.
References
- Suzanne M (2017) Potential therapeutic applications for emu Lipid Technology 29: 28-31.
- Jeengar MK, Kumar SP, Thummuri D, Shrivastava S, Guntuku L, et al. (2015) Review on emu products for use as complementary and alternative Nutrition 31: 21-27.
- Whitehouse MW, Turner AG, Davis CK, Roberts MS (1998) Emu oil (s): A source of non-toxic transdermal anti-inflammatory agents in aboriginal Inflammopharmacology 6: 1-8.
- Beckerbauer LM, Thiel-Cooper R, Ahn DU, Sell JL, Parrish FC, et al. (2001) Influence of two dietary fats on the composition of emu oil and Poultry Science 80: 187-194.
- Howarth G, Lindsay R, Butler R, Geier MS (2008) Can emu oil ameliorate inflammatory disorders affecting the gastrointestinal system?. Australian Journal of Experimental Agriculture 48: 1276-1279.
- Vemu B, Selvasubramanian S, Pandiyan V (2015) Emu oil offers protection in Crohn’s disease model in BMC complementary and alternative medicine 16: 1-9.
- MatlhokoI P, WebbII EC, ChamunorwaI P (2010) Dietary manipulation of oil production in commercial emu. South African Journal of Animal Science 40: 442-445.
- Mashtoub S, Cheah KY, Lansdown N, Howarth G (2014) Emu oil source does not significantly alter therapeutic efficacy, while ostrich oil has no beneficial effect, in a rat model of chemotherapy-induced Journal of Gastroenterology and Hepatology 29: 17.
- Lindsay RJ, Geier MS, Yazbeck R, Butler RN, Howarth GS (2010) Orally administered emu oil decreases acute inflammation and alters selected small intestinal parameters in a rat model of mucositis. British Journal of Nutrition 104: 513-519.
- Bennett DC, Code WE, Godin DV, Cheng KM (2008) Comparison of the antioxidant properties of emu oil with other avian oils. Australian Journal of Experimental Agriculture 48: 1345-1350.
- Snowden JM, Whitehouse MW (1997) Anti-inflammatory activity of emu oils in rats. Inflammopharmacology 5: 127-132.
- Yoganathan S, Nicolosi R, Wilson T, Handelman G, Scollin P, et (2003) Antagonism of croton oil inflammation by topical emu oil in CD‐1 mice. Lipids 38: 603-607.
- Abimosleh SM, Lindsay RJ, Butler RN, Cummins AG, Howarth GS (2012) Emu oil increases colonic crypt depth in a rat model of ulcerative Digestive diseases and sciences 57: 887-896.
- Vahedian V, Asadi A, Esmaeili P, Zamani S, Zamani R, et al. (2020) Anti-inflammatory activity of emu oil-based nanofibrous scaffold through downregulation of IL-1, IL-6, and TNF-α pro-inflammatory cytokines. Hormone molecular biology and clinical investigation 41.
- McLennan P, Howe P, Abeywardena M, Muggli R, Raederstorff D, et al. (1996) The cardiovascular protective role of docosahexaenoic European journal of pharmacology 300: 83-89.
- Calder PC (1997) n-3 polyunsaturated fatty acids and cytokine production in health and disease. Annals of Nutrition and Metabolism 41: 203-234.
- Sethuraman SN, Swaminathan S, Nelson SB, Palaninathan PS, Gopalan TK, et (2015) Modulation of PPARγ and TNFα by emu oil and glycyrrhizin in ulcerative colitis. Inflammopharmacology 23: 47-56.
- Medeiros-de-Moraes IM, Gonçalves-de-Albuquerque CF, Kurz ARM, Jesus Oliveira FM, Pereira de Abreu VH, et (2018) Omega-9 oleic acid, the main compound of olive oil, mitigates inflammation during experimental sepsis. Oxidative medicine and cellular longevity 4: 1-3.
- Arezoumand KS, Alizadeh E, Esmaeillou M, Ghasemi M, Alipour S, et (2018) The emu oil emulsified in egg lecithin and butylated hydroxytoluene enhanced the proliferation, stemness gene expression, and in vitro wound healing of adipose-derived stem cells. In Vitro Cellular & Developmental Biology-Animal 54: 205-216.
- López A, Sims DE, Ablett RF, Skinner RE, Léger LW, et al. (1999) Effect of emu oil on auricular inflammation induced with croton oil in American journal of veterinary research 60: 1558-1561.
- Zanardo V, Giarrizzo D, Volpe F, Giliberti L, Straface G (2017) Emu oilbased lotion effects on neonatal skin barrier during transition from intrauterine to extrauterine life. Clinical, cosmetic and investigational dermatology 10: 299.
- Zanardo V, Giarrizzo D, Maiolo L, Straface G (2016) Efficacy of topical application of emu oil on areola skin barrier in breastfeeding Journal of evidence-based complementary & alternative medicine 21: 10-13.
- Hudson BD, Christiansen E, Tikhonova IG, Grundmann M, Kostenis E, et (2012) Chemically engineering ligand selectivity at the free fatty acid receptor 2 based on pharmacological variation between species orthologs. The FASEB Journal 26: 4951-4965.
- Stoddart LA, Smith NJ, Milligan G (2008) International Union of Phar LXXI. Free fatty acid receptors FFA1,-2, and-3: Pharmacology and pathophysiological functions. Pharmacological reviews 60: 405-417.
- Fujita T, Matsuoka T, Honda T, Kabashima K, Hirata T, et al. (2011) A GPR40 agonist GW9508 suppresses CCL5, CCL17, and CXCL10 induction in keratinocytes and attenuates cutaneous immune inflammation. J Invest Dermatol 131: 1660-1667.
- Nagatake T, Shiogama Y, Inoue A, Kikuta J, Honda T, et al. (2018) The 17,18-epoxyeicosatetraenoic acid-G protein-coupled receptor 40 axis ameliorates contact hypersensitivity by inhibiting neutrophil mobility in mice and cynomolgus macaques. J Allergy Clin Immunol 142: 470 -484.
- Williams HC (2005) Clinical Atopic dermatitis. New England Journal of Medicine 352: 2314-2324.
- Elias PM, Schmuth M (2009) Abnormal skin barrier in the etiopathoge- nesis of atopic dermatitis. Current allergy and asthma reports 9: 265-272.
- Agrawal R, Woodfolk JA (2014) Skin barrier defects in atopic Current allergy and asthma reports 14: 433.
- Simpson EL, Chalmers JR, Hanifin JM, Thomas KS, Cork MJ, et al. (2014) Emollient enhancement of the skin barrier from birth offers effec- tive atopic dermatitis Journal of Allergy and Clinical Immuno- logy 134: 818-823.
- Yamamoto A, Serizawa S, Ito M, Sato Y (1991) Stratum corneum lipid abnormalities in atopic dermatitis. Archives of dermatological research 283: 219-223.
- Saevik BK, Thoresen SI, Taugbøl O (2002) Fatty acid composition of serum lipids in atopic and healthy dogs. Research in veterinary science 73: 153-158.
- Son S, Park S, Koh J, Im D (2020) Free fatty acid receptor 4 (FFA4) ac- tivation ameliorates 2, 4-dinitrochlorobenzene-induced atopic dermatitis by increasing regulatory T cells in mice. Acta Pharmacologica Sinica 41: 1337-1347.
- Piccolo D (2009) The role of Emu Oil in the treatment of Atopical Derma- NPT (New Prospects in Treatment) Journal 2: 1-8.
- Piccolo D (2010) Evaluating the Efficacy and tolerability of Emutopic li- pid gel and cream as a coadjuvant treatment of mild to moderate atopical NPT (New Prospects in Treatment) Journal 3: 2-7.
- Borda LJ, Wikramanayake TC (2015) Seborrheic dermatitis and dandruff: A comprehensive review. Journal of clinical and investigative dermatology.
- DeAngelis YM, Gemmer CM, Kaczvinsky JR, Kenneall DC, Schwartz JR (2005) Three etiologic facets of dandruff and seborrheic dermatitis: Malassezia fungi, sebaceous lipids, and individual sensitivity. Journal of Investigative Dermatology Symposium Proceedings Elsevier 295-297.
- Attarzadeh Y, Asilian A, Shahmoradi Z, Adibi N (2013) Comparing the efficacy of Emu oil with clotrimazole and hydrocortisone in the treatment of seborrheic dermatitis: A clinical trial. Journal of research in medical sciences 18: 477.
- Rollmann DC, Novotny PJ, Petersen IA, Garces YI, Bauer HJ, et (2015) Double-blind, placebo-controlled pilot study of processed ultra emu oil versus placebo in the prevention of radiation dermatitis. International Journal of Radiation Oncology 92: 650-658.
- Penturf M, Banion SO, Griswold JA (1998) Evaluation of emu oil in lubrication and treatment of healed burn wounds. Proceedings of American Burn Association 18.
- Kohl E, Steinbauer J, Landthaler M, Szeimies RM (2011) Skin ageing. Journal of the European academy of dermatology and venereology 25: 873-884.
- Kim EJ, Kim MK, Jin XJ, Oh JH, Kim JE, et (2010) Skin aging and photoaging alter fatty acids composition, including 11, 14, 17-eicosatrienoic acid, in the epidermis of human skin. Journal of Korean medical science 25: 980-983.
- Nicolai E, Sinibaldi F, Sannino G, Laganà G, Basoli F, et (2017) Omega-3 and Omega-6 fatty acids act as inhibitors of the matrix metalloproteinase-2 and matrix metalloproteinase-9 activity. The protein journal 36: 278-285.
- Rogers J, Harding C, Mayo A, Banks J, Rawlings A (1996) Stratum corneum lipids: The effect of ageing and the Archives of dermatological research 288: 765-770.
- Pilkington SM, Watson REB, Nicolaou A, Rhodes LE (2011) Omega‐3 polyunsaturated fatty acids: Photoprotective Experimental dermatology 20: 537-543.
- Brigham E, McCormack MC, Woo H, Rice J, Koehler K, et al. (2018) Omega-3 and omega-6 fatty acid intake modifies response to indoor air pollution in children with A16 Air Pollution and Pediatric Asthma American Thoracic Society A1017-A1017.
- Nejati-Koshki K, Pilehvar-Soltanahmadi Y, Alizadeh E, Ebrahimi-Kalan A, Mortazavi Y, et al. (2017) Development of Emu oil-loaded PCL/ collagen bioactive nanofibers for proliferation and stemness preservation of human adipose-derived stem cells: Possible application in regenerative Drug development and industrial pharmacy 43: 1978-1988.
- Zemtsov A, Gaddis M, Montalvo-Lugo VM (1996) Moisturizing and cosmetic properties of emu oil: A pilot double blind Aust J Dermatology 37: 159-162.
Citation: Milani M (2021) Emu Oil Topical Use in Dermatology. J Clinic Exper Cosme Derma 4: 020.
Copyright: © 2021 Milani M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.


